Regeneron - Trials & Tribulations
A little perspective after a rough week.
Friends of NFTBC.
I don’t check my share prices particularly often - perhaps once every couple of weeks, unless I’m considering some kind of action. But there are occasions when share prices come and check on you, whether you like it or not. May 30th was one such occasion, when Regeneron released the headline results for its two itepekimab phase III trials in COPD.
COPD
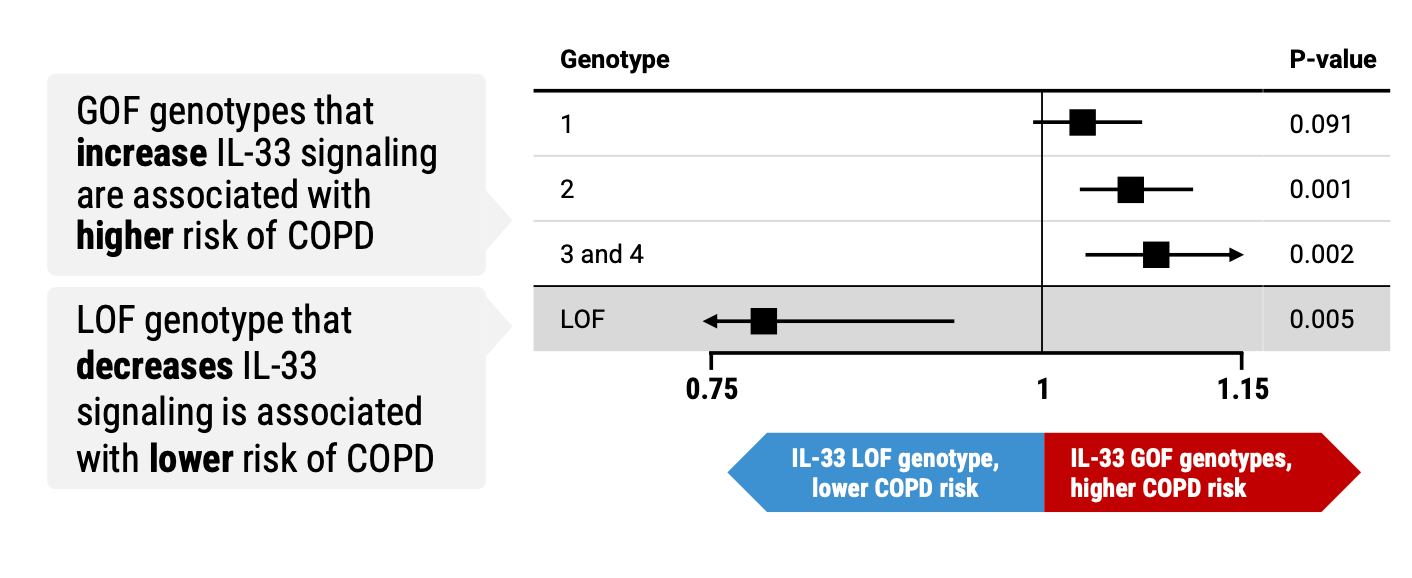
As background, itepekimab (antibody targeting IL-33) had previously demonstrated particularly encouraging results at phase II in COPD patients who were former smokers, with a 42% reduction in acute exacerbations and a p-value of 0.006. Additionally, RGC had discovered a very strong genetic link between IL-33 gain and loss of function and risk of disease:
As such, more or less everyone was anticipating the AERIFY phase III trials to be successful. In the event, one trial met its primary endpoint and one did not:
The effect on the shares was an instant plunge of 18% and a $12bn drop in market cap. Was this justified? My answer may surprise you: Yes, maybe! It just depends which game you’re playing. As noted, everyone was expecting a ‘win’ here (me included). Additionally, REGN is perceived by many to have a poor pipeline1 so they really ‘needed the win’. If a pharma business has little pipeline promise and is failing phase III trials, the outlook could indeed be quite bleak - in these sorts of situations a low multiple of earnings seems fair. Similarly, under industry norms a pharma business is typically expected to buy its way out of trouble through M&A, and so REGN’s monkish abstinence from large deals simply adds fuel to the fire.